The StemLife team supported the organizers and attended the "Short course on Cord Blood Transplantation" meeting held today in Singapore's most prestigious research facility Biopolis (equivalent to the NIH for the lion city). Co-organized by Johns Hopkins, National University Hospital, Singapore Cord Blood Bank (SCBB) and the local Society of Haematology, it was quite a successful turnout of speakers and attendees who provided a lively audience.
Guest speakers today included Prof. Eliane Gluckman (1st cord blood stem cell transplanter), Prof. EJ Schpall (MD Anderson, FACT-Netcord accreditation), Prof. Takahashi of Tokyo University, Prof. CK Li from Prince of Wales Hospital Hong Kong, Prof. Ian McNeice, Johns Hopkins Singapore. Local speakers included Singapore's well known haem-oncologists Dr. Tan Ah Moy, Dr. Tan Poh Lin and Dr. William Hwang (now medical director of SCBB) and Dr. Michael Choi of Viacell Singapore.
Attendees included a collection of representatives of cord blood banks (largest stem cell company turnout from StemLife!), doctors from local hospitals and interested physicians and some students.
The seminars revolved largely around the results of cord blood stem cell transplantation since the first one performed by Prof. Gluckman, it is quite clear that the process of transplantation has been refined and improved tremendously. The data from her talk showed that unrelated donor cord blood stem cell transplants were as good as or better than proceeding with unmatched, unrelated bone marrow transplants. Nucleated cell number is still used as the transplant indicator as the correlation with CD34+ cells in cord blood was still tenuous, and the Gluckman recommendation was that at least 2x10e7 NC/Kg at infusion must be obtained for a greater chance of success in malignant diseases (StemLife derives its cut-off value from this recommendation). A member of a pharmaceutical group from India raised the question if she would use a unit which was contaminated with bacteria, given that it could be treated with antibiotics. Prof. Gluckman answered that she would not accept nor use the unit for fear of an adverse and unpredictable effect, which could lead to the death of the patient.
Also discussed were new protocols in cord blood transplantation using double bags of cord blood stem cells which should be as closely matched as possible and have shown promising results although the procedure still requires refinement. Prof. Gluckman's conclusion was that "cord blood is a unique resource for transplantation, regenerative medicine and scientific research, and for this purpose, there is an increasing need for the number of quality units stored".
Prof. EJ Schpall's talk centred largely on FACT-Netcord Accreditation, which is probably most applicable to the public banks who wish to trade their units internationally. She was quite clear on the fact that "public banks" around the world were in competition to "sell" the most number of units for transplantation (USD 21,000 per unit). A high barrier of entry is created through complying with standards and required protocols. She also stressed that FACT only accepts banks storing in cryobags and do not accept vials or any other form of storage.
This criteria would immediately eliminate banks storing their units in vials from participating in FACT accreditation or wishing to join Netcord in the future.
Friday, March 31, 2006
Wednesday, March 29, 2006
Viewpoint on Stem Cell Banking
When I decided to do a search on Angel, I was thrilled that many blogsites have featured Angel's story. The aim of her parents in sharing the story was to enlighten and encourage other parents in Malaysia and the world to know that stem cells do exist in umbilical cord blood, and that it is possible to use the cells for a matched sibling in need. Her mother also expressed her happiness and satisfaction with StemLife's service and that the stem cells were viable for use.
Given all the medically published information available today, I am still astonished that a few local doctors question if umbilical cord blood stem cells should be collected at all and advise their patients that they will never need to use the stem cells and that banking stem cells would be a "waste of their money".
The number of therapies of stem cells derived from umbilical cord blood has increased since 1987, whilst the prices charged by many private companies for cord blood stem cell banking services have either stayed the same or decreased in price. Either way, clients and patients benefit from a lower price point from the perspective of inflation and an increased access to therapeutics and new research. In the next 15-20 years, it may be possible that stem cells could be used to treat many more conditions than just cancer or blood disorders.
Personally, my take on stem cells are as follows:
1. Stem cells may be useful in the future, bank them if it can be done so at reasonable cost and cause no bodily harm
2. Whether in private or public setting, there are many babies being born on a daily basis and not even 10% per year is being collected and banked in the most developed countries (US has approx. 4 million births per year).
Taking US figures -which would more fairly depict a mature stem cell banking industry- till today less than 10% of the total numbers of babies born have their cord blood stem cells banked privately or publicly. Frankly, I find it curious that public stem cell banks consider themselves as competing in the same space for umbilical cords. Surely, if it were freely accessible, the other 90% of mothers could be given the opportunity to donate their baby's stem cells with the possibility of saving another person's life.
I did some searching and found the possible reasons why this isn't so:
a) It's a combination of the high cost of processing and storing donated blood at public cord blood banks and a lack of awareness about cord blood donation among parents-to-be.
b) it costs a public bank about USD 1,000 to collect, rigorously test, freeze, and store a single unit of cord blood. With a price tag that high, and the public need for cord blood still relatively low (for transplantation in cancers and blood disorders that is), the day when every baby's cord blood is routinely stored probably won't come soon.
c) You aren't likely to see a widely publicized national drive to encourage new cord blood donors either. "Most public banks operate on a shoestring budget," says Mary Halet, cord blood program manager of the National Marrow Donor Program, a nonprofit organization that maintains the largest public listing of umbilical cord blood units available for transplantation in the United States. "With 4 million births annually, the current system would be overburdened if even one-quarter opted for donation." As a result, most public banks work with a select group of collecting hospitals, so they can manage the volume, Halet says.
Parents considering cord blood donation are also encouraged to enquire if their cord blood would be released to them should they need it, and whether their baby's stem cells will be used for research and not for transplantation. Effectively, parents release their rights over the cord blood stem cells to the bank and no longer have any control over its fate.
When choosing a private stem cell bank, quality of service ranks high on the selection factor. No point in selecting a bank which is cheap in pricing when there isn't a committment to ensuring that the cord blood units are collected right, delivered within 24 hrs to the processing laboratory and banked by a team who have the right management skills and professional background. A StemLife policy is that every unit is treated as if it were our own, right from the collection through to its cryopreservation.
A final note: StemLife occasionally receives enquiries about donation and patients needing stem cells, and with no hesitation, we have referred them to the local public authority. Just like public hospitals and private hospitals- a different segment of the population needs to be served. Similar to blood donation and banking, patients undergoing elective surgery may be asked to store their blood with the hospital a week in advance of their elective operation, preserving donated units for emergencies and others in need, stem cells banked privately will less likely drain public resources in future.
Allow patients and clients the right to choose, and public and private banks can co-exist for the betterment of everyone at large.
Given all the medically published information available today, I am still astonished that a few local doctors question if umbilical cord blood stem cells should be collected at all and advise their patients that they will never need to use the stem cells and that banking stem cells would be a "waste of their money".
The number of therapies of stem cells derived from umbilical cord blood has increased since 1987, whilst the prices charged by many private companies for cord blood stem cell banking services have either stayed the same or decreased in price. Either way, clients and patients benefit from a lower price point from the perspective of inflation and an increased access to therapeutics and new research. In the next 15-20 years, it may be possible that stem cells could be used to treat many more conditions than just cancer or blood disorders.
Personally, my take on stem cells are as follows:
1. Stem cells may be useful in the future, bank them if it can be done so at reasonable cost and cause no bodily harm
2. Whether in private or public setting, there are many babies being born on a daily basis and not even 10% per year is being collected and banked in the most developed countries (US has approx. 4 million births per year).
Taking US figures -which would more fairly depict a mature stem cell banking industry- till today less than 10% of the total numbers of babies born have their cord blood stem cells banked privately or publicly. Frankly, I find it curious that public stem cell banks consider themselves as competing in the same space for umbilical cords. Surely, if it were freely accessible, the other 90% of mothers could be given the opportunity to donate their baby's stem cells with the possibility of saving another person's life.
I did some searching and found the possible reasons why this isn't so:
a) It's a combination of the high cost of processing and storing donated blood at public cord blood banks and a lack of awareness about cord blood donation among parents-to-be.
b) it costs a public bank about USD 1,000 to collect, rigorously test, freeze, and store a single unit of cord blood. With a price tag that high, and the public need for cord blood still relatively low (for transplantation in cancers and blood disorders that is), the day when every baby's cord blood is routinely stored probably won't come soon.
c) You aren't likely to see a widely publicized national drive to encourage new cord blood donors either. "Most public banks operate on a shoestring budget," says Mary Halet, cord blood program manager of the National Marrow Donor Program, a nonprofit organization that maintains the largest public listing of umbilical cord blood units available for transplantation in the United States. "With 4 million births annually, the current system would be overburdened if even one-quarter opted for donation." As a result, most public banks work with a select group of collecting hospitals, so they can manage the volume, Halet says.
Parents considering cord blood donation are also encouraged to enquire if their cord blood would be released to them should they need it, and whether their baby's stem cells will be used for research and not for transplantation. Effectively, parents release their rights over the cord blood stem cells to the bank and no longer have any control over its fate.
When choosing a private stem cell bank, quality of service ranks high on the selection factor. No point in selecting a bank which is cheap in pricing when there isn't a committment to ensuring that the cord blood units are collected right, delivered within 24 hrs to the processing laboratory and banked by a team who have the right management skills and professional background. A StemLife policy is that every unit is treated as if it were our own, right from the collection through to its cryopreservation.
A final note: StemLife occasionally receives enquiries about donation and patients needing stem cells, and with no hesitation, we have referred them to the local public authority. Just like public hospitals and private hospitals- a different segment of the population needs to be served. Similar to blood donation and banking, patients undergoing elective surgery may be asked to store their blood with the hospital a week in advance of their elective operation, preserving donated units for emergencies and others in need, stem cells banked privately will less likely drain public resources in future.
Allow patients and clients the right to choose, and public and private banks can co-exist for the betterment of everyone at large.
Labels:
banking,
cord blood,
malaysia,
stem cell transplant,
StemLife
Sunday, March 26, 2006
Olfactory Mucosa Stem Cell Surgery in Portugal for Paralysis
Spinal cord surgery for the implantation of stem cells derived from umbilical cord blood or olfactory mucosa has been performed and announced in a some countries, including Holland, Korea and Portugal. The latest article relates a story, about an 18 year-old former volleyball player from Illinois (paralyzed for 2 years from the waist down after a car accident) who went to Portugal for 12 days to have this procedure, in a fair amount of detail.
The procedure took place at a hospital called Hospital de Egas Moniz and the patient was operated on by a Portuguese neurologist, Dr. Carlos Lima. The surgery is still experimental and involved the removal of tissue from the olfactory bulb, which is thought to contain stem cells that will promote the formation of nerve cells and new neuron connections. The tissue was then inserted into the 4 centimeter damaged area of the patient's spinal cord.
The artice states:"All of Lima's approximately 80 patients - half of them Americans - who have undergone the surgery in Portugal since summer 2002 have reported some gains in sensation, and about 5 percent say they have better feeling in their bladders and bowels, Hinderer said." But according to a Dec 4 report by the Detroit Free Press, It has been noted that most patients do not see any substantial gains until 6 months post surgery.
Additionally, another website states (scroll half way down):"All of the patients recovered well from the surgery. Olfaction returned to normal by three months after the surgery. All of the patients showed improvements. One of the patients regained bladder control at fifteen months after the surgery. Regaining bladder control is extremely important to patients with spinal cord injury. All but one of the patients gained feeling in some areas of their body where they previously had no feeling. All of the patients gained the ability to move certain muscles that they could not move before the olfactory mucosa treatment."
After the operation, the patient had to undergo intensive rehabilitation program for two and a half weeks at a not for profit rehab institute operated by Dr. Steven Hinderer in Michigan. According to Dr. Hinderer, the aggressive excercises, which include crawling and walking with the help of braces for three hours each day, was to frequently stimulate for reorganizing and adapting the body to the changes that take place within.
Critics say the surgery/rehab combination is a waste of money (USD 44,000 for the surgery, USD 10,000 for the rehab, USD 10,000 for the leg brace and USD 5,000 for the work out equipment, total USD 69K) , but neither the patient nor her mother regret the pain or effort connected with the surgery. They believe that the surgery has helped the patient restore feelings in the hips and some sensation in her legs.
"Without the surgery, Jacki probably wouldn't regain any sensation," Her mother said. "There's lots of stuff going on, I'm sure she has more than she had before she went to Portugal." Although spinal cord repair with stem cells is in its experimental stages, this patient who has many years ahead of her is brave enough to accept the risks and at least show that thus far, it hasn't caused her any harm. We'll just have to wait and see if it helps her regain significant control of her muscles. Keep an open mind and support those who wish to improve their lives and help others in similar situations.
Jacki Rabon has written about her experience in Portugal, Detroit and Waverly in an online journal .
The procedure took place at a hospital called Hospital de Egas Moniz and the patient was operated on by a Portuguese neurologist, Dr. Carlos Lima. The surgery is still experimental and involved the removal of tissue from the olfactory bulb, which is thought to contain stem cells that will promote the formation of nerve cells and new neuron connections. The tissue was then inserted into the 4 centimeter damaged area of the patient's spinal cord.
The artice states:"All of Lima's approximately 80 patients - half of them Americans - who have undergone the surgery in Portugal since summer 2002 have reported some gains in sensation, and about 5 percent say they have better feeling in their bladders and bowels, Hinderer said." But according to a Dec 4 report by the Detroit Free Press, It has been noted that most patients do not see any substantial gains until 6 months post surgery.
Additionally, another website states (scroll half way down):"All of the patients recovered well from the surgery. Olfaction returned to normal by three months after the surgery. All of the patients showed improvements. One of the patients regained bladder control at fifteen months after the surgery. Regaining bladder control is extremely important to patients with spinal cord injury. All but one of the patients gained feeling in some areas of their body where they previously had no feeling. All of the patients gained the ability to move certain muscles that they could not move before the olfactory mucosa treatment."
After the operation, the patient had to undergo intensive rehabilitation program for two and a half weeks at a not for profit rehab institute operated by Dr. Steven Hinderer in Michigan. According to Dr. Hinderer, the aggressive excercises, which include crawling and walking with the help of braces for three hours each day, was to frequently stimulate for reorganizing and adapting the body to the changes that take place within.
Critics say the surgery/rehab combination is a waste of money (USD 44,000 for the surgery, USD 10,000 for the rehab, USD 10,000 for the leg brace and USD 5,000 for the work out equipment, total USD 69K) , but neither the patient nor her mother regret the pain or effort connected with the surgery. They believe that the surgery has helped the patient restore feelings in the hips and some sensation in her legs.
"Without the surgery, Jacki probably wouldn't regain any sensation," Her mother said. "There's lots of stuff going on, I'm sure she has more than she had before she went to Portugal." Although spinal cord repair with stem cells is in its experimental stages, this patient who has many years ahead of her is brave enough to accept the risks and at least show that thus far, it hasn't caused her any harm. We'll just have to wait and see if it helps her regain significant control of her muscles. Keep an open mind and support those who wish to improve their lives and help others in similar situations.
Jacki Rabon has written about her experience in Portugal, Detroit and Waverly in an online journal .
Saturday, March 25, 2006
New R&D addressing Stem Cell Rejection

I mentioned previously how this whole new industry being built on stem cells would lead to interesting and possibly lucrative spin-offs with impact on lives everywhere. This article mentions a possible example.
Norwood Abbey, an Austalian company which has made several key acquisitions in the United States, has announced that its immunology subsidiary has signed a tripartite research agreement with Monash University (Melbourne) and the Australian Stem Cell Centre to focus on developing methods of controlling the immune system to minimize rejection of stem cell therapies.
The article states:"Under the agreement, Norwood Immunology will provide the ASCC with access to its thymic and bone marrow regrowth technology. This provides a method of replenishing stem cells and potentially facilitates the engraftment and uptake of introduced stem cells into the body, improving their ability to repair damaged tissue and minimizing their likelihood of rejection."
CAPITAL$$
The research will be funded jointly by the Australian Stem Cell Centre and Norwood Immunology and the intellectual property that results from the work will be jointly commercialized by both parties. It would make sense if Norwood's contribution were to be in time and materials (and possibly a bit of cash). This seems to be about stem cell expansion (and perhaps some immune modulation drugs?).
If a company could identify a way in which the immune system could be tightly regulated to accept foreign cells(very complex biocehmistry!), this would revolutionize stem cell therapy. No more HLA matching, less or no drugs, resulting in less side effects. It would be really meaningful for cancer patients and others undergoing stem cell transplants.
Every collaboration like this adds fuel to the fire and somewhere in the world, more dollars are spent on the conviction of stem cell therapy everyday.
Thursday, March 23, 2006
100 Companies Developing Human Stem Cell Therapies
At a recent stem cell conference in San Francisco, the host, a venture capitalist from a reknown Life Science VC (controlling more than USD 500 million in funds) estimated that about 100 companies in the United States and approximately the same number in other countries were trying to develop stem cell based therapies and related products.
Though commercial blockbusters rivaling the sale of drugs is still years away, its nice to know that many of the CEOs from the stem cell companies who attended were optimistic that stem cells are no longer a theory but soon to be reality in the treatment of certain diseases and degenerative conditions.
PICK OF THE CROP (as mentioned in article):
1. Osiris Therapeutics of Baltimore- allogeneic (donated cells) cellular repair for damaged hearts and arthritic knees
2. StemCells of Palo Alto- neural stem cell treatment from fetuses for Batten disease (fatal, caused by missing/defective brain enzyme)
3. Advanced Cell Technology of Alameda- stem cells to treat eye macular degeneration and skin wound healing
4. Geron of Menlo Park- human embryonic stem cells for human spinal cord injury (will be the 1st federally approved embryonic stem cell treatment test)
5. Aastrom Biosciences of Michigan- stem cell treatment for regenerating severe bone fractures
CAPITAL $$
"That's our No.1 challenge, access to capital" said the CEO of Vistagen. Stem cell research does not come cheap and Osiris estimated that they have spent more than USD 200 million on stem cell studies and USD 48 million more this year. With the lack of government funding and the on-going debate on stem cells, many of the companies are finding it hard to raise investor interests to contribute to efforts to ease human suffering with stem cell therapy, and a chance to derive a decent return on investment.
In a separate press release, Samaritan Pharmaceuticals has developed a drug, currently progressing through pre-clinical trials, named SP-04 which induces dormant brain neuronal cells to differentiate rapidly into adult neuron cells for Alzheimer's. The share price has moved 76% on the announcement.
In any industry, the lack of funds will naturally exclude any participants without adequate resources to see the project through. It will be interesting to see the M&A activities that the stem cell industry will generate on the global stage in time to come. The merchant banks are probably getting ready.
Though commercial blockbusters rivaling the sale of drugs is still years away, its nice to know that many of the CEOs from the stem cell companies who attended were optimistic that stem cells are no longer a theory but soon to be reality in the treatment of certain diseases and degenerative conditions.
PICK OF THE CROP (as mentioned in article):
1. Osiris Therapeutics of Baltimore- allogeneic (donated cells) cellular repair for damaged hearts and arthritic knees
2. StemCells of Palo Alto- neural stem cell treatment from fetuses for Batten disease (fatal, caused by missing/defective brain enzyme)
3. Advanced Cell Technology of Alameda- stem cells to treat eye macular degeneration and skin wound healing
4. Geron of Menlo Park- human embryonic stem cells for human spinal cord injury (will be the 1st federally approved embryonic stem cell treatment test)
5. Aastrom Biosciences of Michigan- stem cell treatment for regenerating severe bone fractures
CAPITAL $$
"That's our No.1 challenge, access to capital" said the CEO of Vistagen. Stem cell research does not come cheap and Osiris estimated that they have spent more than USD 200 million on stem cell studies and USD 48 million more this year. With the lack of government funding and the on-going debate on stem cells, many of the companies are finding it hard to raise investor interests to contribute to efforts to ease human suffering with stem cell therapy, and a chance to derive a decent return on investment.
In a separate press release, Samaritan Pharmaceuticals has developed a drug, currently progressing through pre-clinical trials, named SP-04 which induces dormant brain neuronal cells to differentiate rapidly into adult neuron cells for Alzheimer's. The share price has moved 76% on the announcement.
In any industry, the lack of funds will naturally exclude any participants without adequate resources to see the project through. It will be interesting to see the M&A activities that the stem cell industry will generate on the global stage in time to come. The merchant banks are probably getting ready.
Awesome video describing what happens in a heart attack
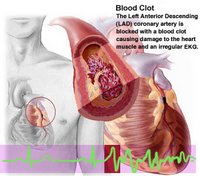
I found this nice video online that describes heart attacks in a simple manner that is very easy to understand. Scroll half way down the page and look for a little square box that you have to click on in order to launch the video player. It only lasts 1 min 50 seconds so it isn't too boring. 3D animation with clear narration... fantastic!
According to the website, "The American Heart Association estimates that in 2004, more than 1 million Americans will suffer a heart attack. About 700,000 of these will be first–time heart attack sufferers, while about 500,000 will be people who had previously had a heart attack." Its a big problem worldwide, Malaysia no exception. It would be great if somehow stem cells could be used in the treatment of its ill after-effects, combined with right medication.... and a proper diet of course.
Wednesday, March 22, 2006
Machines Made to Measure Adult Stem Cell Potency in the Heart
With many doctors still unconvinced that stem cell therapy for the heart amounts to anything more than mere patient "feel good" psychology and the jury still out on the number of stem cells required for optimal results, Cordis- a Johnson & Johnson subsidiary company- is paving the way for the pioneers to prove that it works.
At the 2006 American College of Cardiology Scientific Session, doctors from Vienna Medical Centre in Austria established that patients who received more stem cells experienced a higher degree of cardiac improvement. Although the doctor cautiously said that more work needs to be done, he does admit that this news is encouraging for patients who suffer from serious heart disease.
The improvements after either intramyocardial injection (direct into heart muscle) or intra-coronary injection (like the angioplasty procedure) after six months found a 4.7% increase in patient's heart muscle contractions. Interestingly, the patients also experienced a 4.9% decrease in blood flow problems upon receipt of a stress inducing drug -I'm not exactly sure what this means but I'll find out.
Cordis announced in a press release that they have come up with 2 supporting products for stem cell application- one is the NOGA system which is a high-tech cardiac navigation system to take precise pictures of the heart; the second is a funky named "Myostar injection catheter" although I am not sure what additional benefits it may have over existing catheters for the delivery of stem cells.
This field has a long way to go and its good that large caps are throwing their hat into supporting revolutionary medical treatments -foreseeing that these procedures may become commonplace in the future- and that they want to have a big part in it.
Tuesday, March 21, 2006
What is GCSF and its relevance to Stem Cells?

A fellow blogger asked me what GCSF is in a previous entry. Sorry I didn't clarify before, please see my explanation below.
GCSF = human Granulocyte Colony Stimulating Factor (several manufacturers but product marketed by Amgen is Neupogen*)
GCSF is a growth factor or a cytokine (body's signal) which is produced naturally in the human body by different tissues to stimulate the bone marrow to produce white blood cells. In the process, blood progenitor cells are also mobilized (moved) into the circulating blood stream.
It is already well understood that mobilized blood progenitor cells are able to engraft (stick to marrow and re- grow) rapidy in patients given high dose chemotherapy and thus restore the blood system in its entirety. These mobilized stem cells have been shown to be transplantable and maintain their function with the same proliferative potential as stem cells from the bone marrow.
Thus, instead of bone marrow harvesting (requires operation suite) to collect stem cells, patients can now undergo stimulation by GCSF and harvest their stem cells safely via an arm vein. This has enabled much greater convenience for stem cell donors worldwide to collect stem cells for their own present (or future use) or to donate to someone in need.
*NEUPOGEN® is indicated for the mobilization of hematopoietic progenitor cells into the peripheral blood for collection by leukapheresis. Mobilization allows for the collection of increased numbers of progenitor cells capable of engraftment compared with collection by leukapheresis without mobilization or bone marrow harvest. After myeloablative chemotherapy‚ the transplantation of an increased number of progenitor cells can lead to more rapid engraftment‚ which may result in a decreased need for supportive care.
First Cord Blood Stem Cell Transplant by StemLife

Malaysia's first cord blood stem cell transplant from a privately banked unit at StemLife was reported on the front page of a local daily newspaper. As a private stem cell bank, we have a few skeptics who question whether our laboratory standards are trustworthy and if the stem cells can be used for transplant. StemLife is very proud to say that not only are we undergoing external accreditation and run by a professional and experienced lab director, but stem cells banked by us are viable and have already been successfully used in a full marrow transplant setting.
Angel Lai is StemLife's first cord blood stem cell patient, her brother Samuel donated his stem cell unit to save her life. Its been almost two years since her transplant and she has since grown tall, strong and is a bundle of lively energy.
I'm proud to share with you that the little girl is doing very well, she's a fighter and doing so well. Her parents are overjoyed and so is the StemLife team! :) We are committed to providing friendly, affordable and quality services to our clients.
Thursday, March 16, 2006
Menstrual Blood Stem Cells- great if you're female?
 What a wonderfully weird article from Japanese researchers on stem cells to brighten up one's day. At the American College of Cardiology, doctors from the Keio University School of Medicine in Tokyo announced the collection of menstrual blood from six women and harvested the stem cells that originated in the lining of the uterus, obtaining 30x more stem cells than bone marrow. In taking this very interesting approach of attempting to convert "waste" to "wealth", I wonder if the harvesting of these cells could be done in a sterile, non-contaminated manner.
What a wonderfully weird article from Japanese researchers on stem cells to brighten up one's day. At the American College of Cardiology, doctors from the Keio University School of Medicine in Tokyo announced the collection of menstrual blood from six women and harvested the stem cells that originated in the lining of the uterus, obtaining 30x more stem cells than bone marrow. In taking this very interesting approach of attempting to convert "waste" to "wealth", I wonder if the harvesting of these cells could be done in a sterile, non-contaminated manner. With all the existing challenges in ensuring a clean collection of cord blood stem cells, and taking an angle from a standpoint of storing and potentially using these cells in therapy as claimed in the article, I wonder if there needs to be a special contraption to collect the stem cells directly from the lining or perhaps at the cervix itself to prevent the usual flora from contaminating the collected unit. On the otherhand, the opportunity to collect it would be once a month! (So if the collection fails this month, give it another go next month...?)
Furthermore, the stem cells were cultured to induce cardiac cell formation and demonstrated contractile ability after 5 days. I thought this was quite a short time, but without reading the full article, its hard to draw any conclusions.
The researcher also emphasized the point that cells obtained from a younger patients would have a longer lifespan than cells harvested from older donors. This is the same reasoning as banking stem cells from cord blood of newborns and choosing a younger donor for blood stem cells. It would be interesting to see if this source of stem cells can be safely banked and used as a viable source of transplantation.
Wednesday, March 15, 2006
Safe Stimulation of Stem Cells by GCSF and Vessel Maintenance

I am often asked if the administration of GCSF is safe. Well, lots of stem cell donors undergo peripheral blood stem cell stimulation and harvesting for donation to a loved one and so far, no adverse reactions have been reported or observed (numbers are estimated to be >40,000 worldwide every year).
It was significant that the GCSF heart trial took place in Germany (see previous entry-Stem Cells for Heart Disease) and that other similar trials are also taking place in other institutions (Baxter trial, Schaer trial) and that although the stimulation of stem cells didn't improve their condition, it certainly did not make them any worse off.
Just a thought- It's already known that GCSF levels in the body rise in response to tissue damage and that stem cells probably need to be administered to site of damage- do circulating stem cells help in maintaining the rest of the peripheral blood vessels protecting them as best they can as long as no congestion or sustained disease is impeding its work?
Under normal circumstances of wear and tear (blood does flow at pretty high pressures near the heart, and like piping, needs regular maintenance), circulating stem cells might play a role in the upkeep of these vessels. Like a traveling sentinel that keeps things in check. I wonder if anyone out there might be thinking of how to devise a study on this...
Sunday, March 12, 2006
StemLife collaborating with Doctors for Heart Stem Cell Therapy in Malaysia
 I'm delighted to share that one of StemLife's collaborating doctors has announced the preliminary results of stem cell therapy for the failing heart in a local Malaysian newspaper.
I'm delighted to share that one of StemLife's collaborating doctors has announced the preliminary results of stem cell therapy for the failing heart in a local Malaysian newspaper.Essentially, what we were doing was to marry two existing techniques from different medical applications. The first is angioplasty (common in heart patients, this involves the insertion of a catheter through blood vessels of the heart and using a balloon to open a blocked artery) and the harvesting of stem cells via the peripheral vein known as apheresis, routinely performed in many major hospitals for cancer patient treatment worldwide.
The stem cells which were harvested from a blood vessel in the arm, is then returned to a blood vessel in the heart. The stem cells are capable of forming new blood vessels thus provide better support for the heart muscles. This cell conversion ability is thought to be the reason for improved blood flow and restoration of the heart's pumping action (ejection fraction).
What I'd like to point out though, is that although StemLife is the first stem cell company to facilitate this procedure in Malaysia, this procedure and concept is not our own. We've been following stem cell therapy for the heart since we started and the first news that got us quite interested was this article describing peripheral blood stem cell use in heart damage in a young man.
Now, this procedure has been documented in reknown heart institutions in many countries, and patients with heart disease have another option should their current therapies fail to improve their condition or quality of life. Our work is well placed with the Baxter trial that I mentioned in my previous posting. One last thing, although the procedure is pretty safe, the preparation of the cells is important- at StemLife, we've worked out the protocols and are now ready to offer patients the opportunity to bank and use their stem cells for their heart if they wish to.
The Malaysian Star paper carried a follow up article in today's publication. Our work is just beginning and I'll provide more information as we go along. :)
Saturday, March 11, 2006
Isolation of stem cells from peripheral blood for heart disease
Baxter is funding a clinical trial for stem cells isolated from the peripheral vein for re-infusion into the heart vessels. I think this is a convenient way of collecting stem cells and hope that this will study will show good results.
Of course, if the results are positive, this will be a huge way forward for Baxter to sell its machines to a new set of medical centres and expand their services to patients. The costs of apheresis are not small and if the volume of patients undergoing this procedure increases, this could help make prices more affordable all around, including cancer patients who are the primary users of this procedure now.
Of course, if the results are positive, this will be a huge way forward for Baxter to sell its machines to a new set of medical centres and expand their services to patients. The costs of apheresis are not small and if the volume of patients undergoing this procedure increases, this could help make prices more affordable all around, including cancer patients who are the primary users of this procedure now.
Monday, March 06, 2006
Allogeneic IV infused Stem Cells for Heart Attack Patients
Following on from one of my previous entries on circulating stem cells' ineffectiveness for heart disease, another study is now underway to see if stem cells obtained from a healthy donor will help patients who have suffered a heart attack.
Essentially, no one knows if this procedure will work any better than the mobilized autologous (patient's own) stem cells and one of the clinicians, Dr. Gary Schaer admits that this technology is still in its infancy. However, 16 centres are involved in the study- which is a lot!- and the results could be very interesting even if it turned out negative (but the fact that they are conducting it... are they onto something already?)
The stem cells they obtain will be matched to the donor and are not expected to trigger a rejection response. They have enrolled 54 patients into the study, where 36 patients will receive stem cells and the remaining 18 will receive a placebo. Each patient will receive 5 million stem cells for each Kg of body weight- this is an interesting number, its about the same number that oncologists give for an autologous transplant for leukemia. The major difference here though, is that it is unclear whether any drugs will be administered prior or during the treatment process.
I'm wondering why allogeneic stem cells? Is this to alleviate stress on collecting from the same patient who has the condition? Or maybe paving the way that younger/healthy donor cells, harvested at a time of no illness may yield positive results?
Let's hope that we'll have some good news on the first volunteer, 76 year-old Rev. Eugene Carter of Elgin who received the first infusion eight days after his heart attack. Keep a lookout for the donor's age.
Essentially, no one knows if this procedure will work any better than the mobilized autologous (patient's own) stem cells and one of the clinicians, Dr. Gary Schaer admits that this technology is still in its infancy. However, 16 centres are involved in the study- which is a lot!- and the results could be very interesting even if it turned out negative (but the fact that they are conducting it... are they onto something already?)
The stem cells they obtain will be matched to the donor and are not expected to trigger a rejection response. They have enrolled 54 patients into the study, where 36 patients will receive stem cells and the remaining 18 will receive a placebo. Each patient will receive 5 million stem cells for each Kg of body weight- this is an interesting number, its about the same number that oncologists give for an autologous transplant for leukemia. The major difference here though, is that it is unclear whether any drugs will be administered prior or during the treatment process.
I'm wondering why allogeneic stem cells? Is this to alleviate stress on collecting from the same patient who has the condition? Or maybe paving the way that younger/healthy donor cells, harvested at a time of no illness may yield positive results?
Let's hope that we'll have some good news on the first volunteer, 76 year-old Rev. Eugene Carter of Elgin who received the first infusion eight days after his heart attack. Keep a lookout for the donor's age.
Saturday, March 04, 2006
Cord Blood Stem Cells for mending the Heart
StemLife cord blood stem cell bankers take note! "In the future this could be used for the treatment of cardiovascular disease" said the leader of the project Prof. Biagio Saitta. Scientists are now researching how cord blood stem cells might be used in the treatment of heart diease. The Coriell Institute has obtained a USD 300,000 grant from the state of New Jersey to explore how cord blood stem cells respond to muscle cells in a low oxygen environment, conditions similar to a heart attack.
The grant was awarded by the New Jersey Commission on Science and Technology and will support research between Coriell Insitute and Cooper University Hospital to elucidate a phenomenon that was first observed at Johns Hopkins University. Stem cells exposed to damaged heart muscle cells didn't themselves become heart cells but still helped reduce swelling and maintain regular rhythm. This finding in itself could lead to a major breakthrough in complementing existing treatments for heart disease.
Their study will focus on genetic changes in a microscopic number of stem cells when they receive little oxygen, a mirror of conditions in damaged portions of a heart. The stem cells will also be observed interacting with rat muscle cells and fibroblasts, which create the collagen that gives organs their shape.
In previous publications, clinicians and scientists have documented the use of peripheral blood and marrow derived stem cells for treating heart disease. Stem cells derived from these sources are autologous (patient's own) and have demonstrated in an number of patients, improvement in the ejection fraction of the heart. If this study yields positive findings, cord blood stem cells may also establish itself as a viable and easily available source.
It would be interesting to have the results of this 2 year study which may start publishing their findings as early as 6-9 mths time. I wonder if the cord blood stem cells need to be matched and how many stem cells will be required to generate the beneficial effect...? I'll be looking out for the announcements.
The grant was awarded by the New Jersey Commission on Science and Technology and will support research between Coriell Insitute and Cooper University Hospital to elucidate a phenomenon that was first observed at Johns Hopkins University. Stem cells exposed to damaged heart muscle cells didn't themselves become heart cells but still helped reduce swelling and maintain regular rhythm. This finding in itself could lead to a major breakthrough in complementing existing treatments for heart disease.
Their study will focus on genetic changes in a microscopic number of stem cells when they receive little oxygen, a mirror of conditions in damaged portions of a heart. The stem cells will also be observed interacting with rat muscle cells and fibroblasts, which create the collagen that gives organs their shape.
In previous publications, clinicians and scientists have documented the use of peripheral blood and marrow derived stem cells for treating heart disease. Stem cells derived from these sources are autologous (patient's own) and have demonstrated in an number of patients, improvement in the ejection fraction of the heart. If this study yields positive findings, cord blood stem cells may also establish itself as a viable and easily available source.
It would be interesting to have the results of this 2 year study which may start publishing their findings as early as 6-9 mths time. I wonder if the cord blood stem cells need to be matched and how many stem cells will be required to generate the beneficial effect...? I'll be looking out for the announcements.
Wednesday, March 01, 2006
Stem Cells for Heart Disease- How? What? When? Where?
The New York Times carried an article on a report from German researchers in the Journal of American Medical Association. It mentioned "the new treatment entailed injecting the heart attack patients with a hormone that induces the bone marrow to send blood-making cells out into the blood stream" as experiments with animals had suggested that blood-making cells would home in on damaged heart tissue, transform into heart muscles cells and rebuild the heart.
The results however, suggest that the "radical" new treatment (new to heart disease but not new to other diseases, will explain in next entry) was tested under rigorous conditions and was found to be "wholly ineffective". The concept that cells are able to home in on the degenerating tissue and elicit new growth seems plausible but for a few problems which I can think of here.
Firstly, the number of stem cells that are mobilized and leave the marrow constitute only a small percentage of the circulation. It would be a few times higher in concentration, but this may not be sufficient to initiate any migration of cells from the fast moving blood stream into the areas of damage.
Secondly, the area of damage may already have poor circulation, given that the "roads" that bring the cells to the site of injury are either congested or destroyed, this will further limit the number of stem cells that will reach the area to exert any form of effectiveness.
Thirdly, mobilized stem cells do not stay for long in the bloodstream and after stimulation has ended, stem cells home in the bone marrow to which they return or differentiate to other cells which are needed (presumably the cells will then have a slightly higher turnover rate). This homing behavior will also limit the stem cells in circulation and reduce the numbers of cells flowing near the site of damage.
In other published articles where signs of improvement after infusion of stem cells has been demonstrated, the stem cells are injected either into the vessel near the damaged area or directly into the damaged area itself. The cells are thought to migrate transendothelially into the damaged muscle from the vessel or act in coordination with the surrounding tissue to encourage angiogenesis (formation of new blood vessels).
StemLife has been able to successfully reproduce the result first shown by Japanese doctors for Peripheral Artery Disease and helped patients with foot ulcers who refused amputation. Furthermore, StemLife doctors have also seen improvements in patients who have resorted to stem cell therapy for heart failure due to a lack of other options- and have also been able to observe similar results to those already published in the US and other reknown heart institutes.
A lot more work needs to be done to understand exactly what processes take place and whether it can be extended to more patients with less severe symptoms. It would be wonderful to be able to use stem cells to slow down the wear and tear of the organs that we depend on the most.
I am most encouraged by the outcomes and our patients delight in their improved quality of life makes our work all the more exciting and worthwhile. In future entries, I shall describe what we have done in a bit more detail so check back for updates! :)
The results however, suggest that the "radical" new treatment (new to heart disease but not new to other diseases, will explain in next entry) was tested under rigorous conditions and was found to be "wholly ineffective". The concept that cells are able to home in on the degenerating tissue and elicit new growth seems plausible but for a few problems which I can think of here.
Firstly, the number of stem cells that are mobilized and leave the marrow constitute only a small percentage of the circulation. It would be a few times higher in concentration, but this may not be sufficient to initiate any migration of cells from the fast moving blood stream into the areas of damage.
Secondly, the area of damage may already have poor circulation, given that the "roads" that bring the cells to the site of injury are either congested or destroyed, this will further limit the number of stem cells that will reach the area to exert any form of effectiveness.
Thirdly, mobilized stem cells do not stay for long in the bloodstream and after stimulation has ended, stem cells home in the bone marrow to which they return or differentiate to other cells which are needed (presumably the cells will then have a slightly higher turnover rate). This homing behavior will also limit the stem cells in circulation and reduce the numbers of cells flowing near the site of damage.
In other published articles where signs of improvement after infusion of stem cells has been demonstrated, the stem cells are injected either into the vessel near the damaged area or directly into the damaged area itself. The cells are thought to migrate transendothelially into the damaged muscle from the vessel or act in coordination with the surrounding tissue to encourage angiogenesis (formation of new blood vessels).
StemLife has been able to successfully reproduce the result first shown by Japanese doctors for Peripheral Artery Disease and helped patients with foot ulcers who refused amputation. Furthermore, StemLife doctors have also seen improvements in patients who have resorted to stem cell therapy for heart failure due to a lack of other options- and have also been able to observe similar results to those already published in the US and other reknown heart institutes.
A lot more work needs to be done to understand exactly what processes take place and whether it can be extended to more patients with less severe symptoms. It would be wonderful to be able to use stem cells to slow down the wear and tear of the organs that we depend on the most.
I am most encouraged by the outcomes and our patients delight in their improved quality of life makes our work all the more exciting and worthwhile. In future entries, I shall describe what we have done in a bit more detail so check back for updates! :)
Subscribe to:
Comments (Atom)



